|
This question has the same explanation as the selective protonation of the carbonyl oxygen in the Fischer esterification or, in general, its stronger Lewis base character. what were looking at for carboxylic acid derivates is the ability of the functional group to stabilize a positive charge on the carbonyl carbon by donating electrons, essentially transferring the positive charge from the carbonyl carbon to the functional group, decreasing reactivity. The arrow in the first step can also start from a lone pair of the double-bonded oxygen of the carbonyl:Īnd you may ask – why not from the oxygen of the other oxygen? Can the hydroxyl group attack the thionyl chloride instead of the carbonyl oxygen? Salt Formation Because of their enhanced acidity, carboxylic acids react with bases to form ionic salts, as shown in the following equations. Sodium pyrrolidone carboxylic acid is a sodium salt of 2 pyrrolidone carbonic acid and derivative of amino acid These ammonium salts are soluble in water and they show ionic characteristics Salts of carboxylic acids formed with heavy metal ions such as Ca+2, Mg+2, Zn+2, Al+3 tend to be relatively water insoluble Shake both tubes, observe. generally an increase in resonance an increase in stability an decrease in reactivity. In the bases of this transformation is the conversion of the OH group into a good leaving.įor example, thionyl chloride reacts with carboxylic acids forming a highly reactive intermediate capable of losing excellent leaving groups HCl and SO 2 upon a nucleophilic addition of a chloride ion (recall the reaction of alcohols with thionyl chloride):  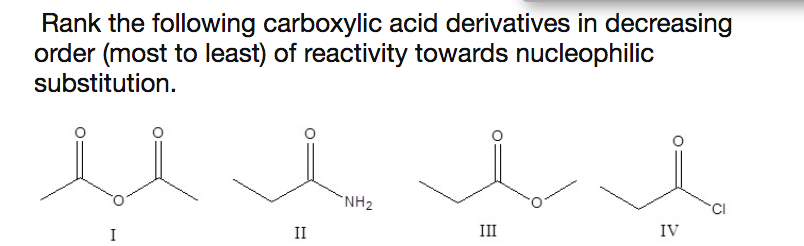
Acyl chlorides can be prepared by reacting carboxylic acids with thionyl chloride (SOCl 2), phosphorous trichloride ( PCl 3) or phosphorous pentachloride ( PCl 5): The amides are the least reactive carboxylic acid derivative because they exhibit substantial carbon-nitrogen double bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
